 
Think of the adverse event as an injured person-bandaging a wound will stop the bleeding, but will not heal the cut. Without those two functions, corrective action merely addresses surface problems, but doesn’t go any deeper to find the systemic cause. 
A weak corrective action system is often due to a fault in one of the two main components: the root cause investigation and effectiveness check. 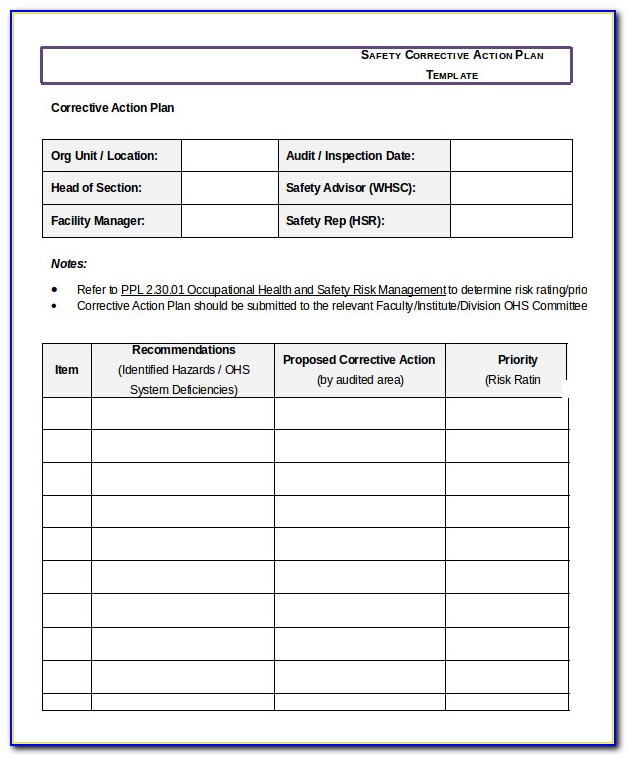
The same or similar issues repeatedly coming up is usually an indication of a problem in the corrective action process. Recurring Issues: What Causes Repeat Problems? Therefore, CAPA has shifted to corrective action under ISO 9001:2015 and will be referred to as such in most cases. 
This definition better aligns with new standards, as “preventive action” has been moved to a risk management function. More simply, it is an evaluation and complete elimination of all risks during product development phases. Preventive action aims to eliminate the occurrence of an event in future products by evaluating product and process designs and controls. This may include addressing the issue identified, determining the scope of the issue, containing the affected product, and correcting the immediate problem. Some people think these are interchangeable, but they are in fact slightly different.Ĭorrective action aims to immediately correct the issue and identify how to prevent a recurrence. This is where organizations need to implement either corrective action or Corrective and Preventive Action (CAPA). When there is a deviation, nonconformance, or other problem, what is done next makes the difference between a quick resolution and a full-blown recall or injured patients. CAPA: What’s the Difference?ĭespite precautions and trainings, issues can (and probably will) still happen. Here, we’ll examine two major components of corrective action systems-root cause investigation and effectiveness check-and how to strengthen them to optimize processes.Ĭorrective Action vs. But it’s not enough to just have one-it must be efficient and effective enough to handle issues in a fast-paced and high-impact industry. ISO standards and industry regulations require medical device organizations to have a corrective action process. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
